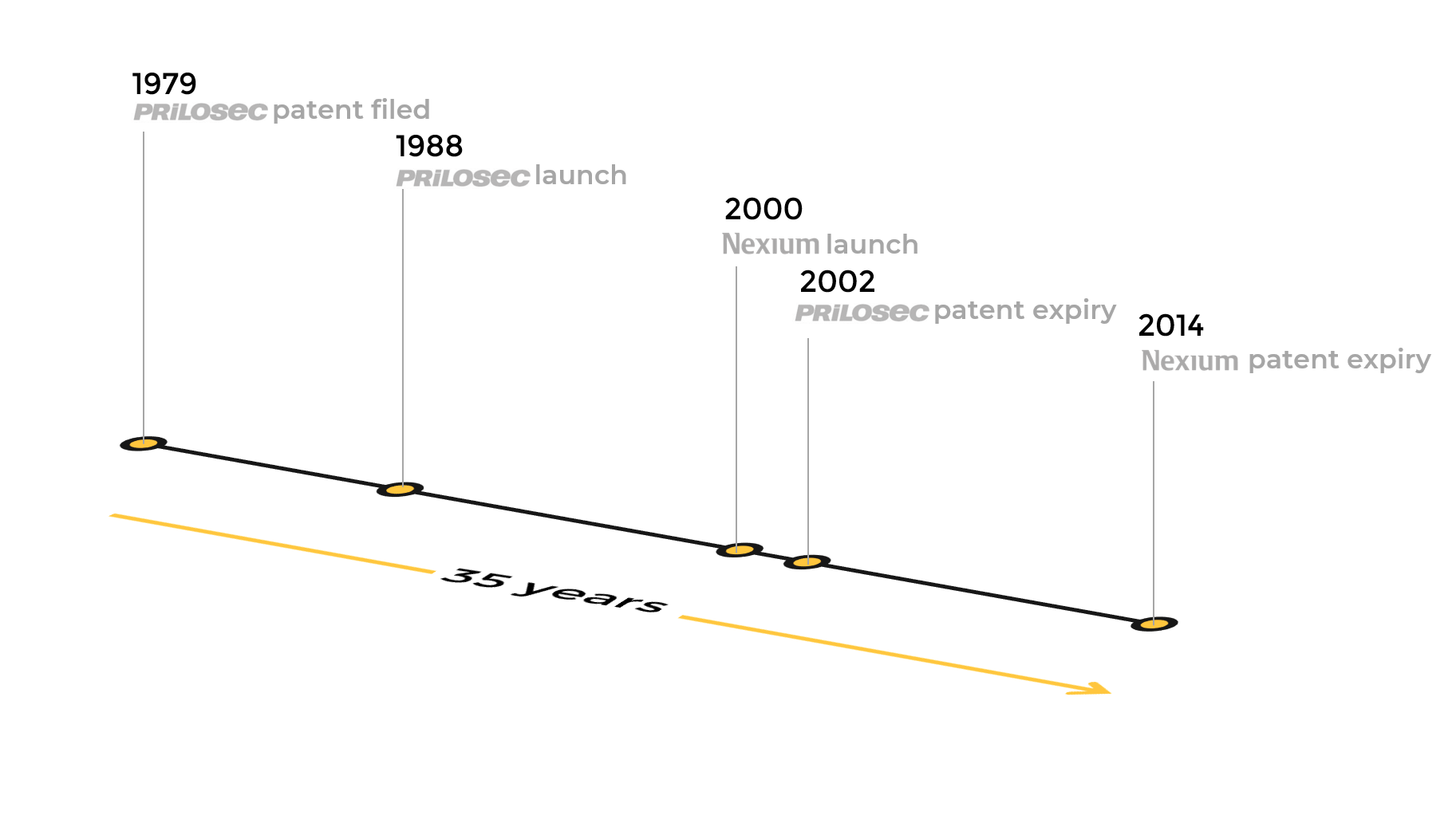
AstraZeneca filed the the first patent of omeprazole in 1979. After 9 years, Prilosec was launched to the market. By 1996, Prilosec became the world’s best-selling drug and in 2000 its annual sales reached $6.3 billion . The problem was that Prilosec's patent should have expired in late 2002. So AstraZeneca planned to develop and launch a next generation drug to maintain the success in the market.
Nexium, the next generation drug developed from Prilosec, proved more effective in treating patients. AstraZeneca submitted Nexium file to the FDA early enough to assure its approval before the patent expiration of Prilosec. In 2000, AstraZeneca launched Nexium before the patent expiration of Prilosec.
The company transferred 40% of Prilosec patients to next-generation Nexium as well as managing a 9 per cent growth in its gastrointestinal franchise in 2001 alone.

The shift from Prilosec to Nexium is a successful implementation of Evergreening strategy.
The patent protection for Nexium, which was an improvement over Prilosec, expired in 2014, this means 35 years after the original patent for Prilosec was taken out.